The water around proteins absorbs more terahertz radiation than pure water, which helps our proteins fold correctly.
If unfolded or misfolded proteins start to accumulate, the endoplasmic reticulum becomes stressed and activates a signaling pathway called the unfolded protein response.
The unfolded protein response is implicated in many diseases. For example, the response is often activated in rapidly growing cancer cells, which enables the cells to fold the large numbers of mutated proteins they produce. Also, viruses can trigger this response as part of their strategy to trick host cells into producing a number of difficult to fold viral proteins.
The correct folding of proteins is essential for cellular homeostasis and the prevention of disease. During times of stress or changes in demand for newly synthesized proteins, signaling pathways cooperate with networks of protein-folding and protein-degradation factors to maintain the pools of non-native proteins within a tolerable range. Such events are crucial for survival, as aberrant protein homeostasis can result in diseases that range from cancer and diabetes to neurodegeneration.
Understanding molecular vibrations in proteins are important because they regulate the function of proteins, e.g., in enzyme catalysis and protein interactions. In particular, low-frequency collective vibrational modes of proteins in the terahertz frequency region are expected to have a strong influence on protein function because terahertz radiation is absorbed by proteins.
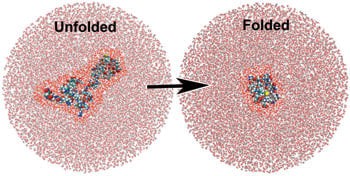
Image: A simulation showing water molecules around an unfolded and folded protein. The water molecules closest to the protein are highlighted bright red for effect. Terahertz spectroscopy has shown that the water molecules closest to the protein behave differently than the surrounding ones and actually participate in the folding process.

fenofibrate 160mg cost tricor 200mg generic fenofibrate online buy
cost ketotifen geodon without prescription tofranil 75mg brand
buy tadalafil 10mg without prescription order sildenafil pill viagra 50mg cheap
oral minoxytop mintop brand buy ed medication
buy acarbose 50mg generic fulvicin online order griseofulvin 250mg for sale
aspirin sale zovirax cream zovirax medication
order dipyridamole 100mg for sale felodipine pill pravastatin 10mg without prescription
order melatonin generic order aygestin generic capsules danazol 100mg
purchase duphaston without prescription order dapagliflozin 10 mg for sale order jardiance 10mg pills
Agora, a tecnologia de posicionamento tem sido amplamente utilizada. Muitos carros e telefones celulares têm funções de posicionamento e também existem muitos aplicativos de posicionamento. Quando seu telefone for perdido, você pode usar essas ferramentas para iniciar rapidamente as solicitações de rastreamento de localização. Entenda como localizar a localização do telefone, como localizar o telefone depois que ele for perdido?
Desde que haja uma rede, a gravação remota em tempo real pode ser realizada sem instalação de hardware especial.
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article.
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article.
Hello there, just became alert to your blog through Google, and found that it is really informative.
I am going to watch out for brussels. I’ll be grateful if
you continue this in future. Many people will be benefited from your writing.
Cheers!
Very interesting topic, thanks for posting.?
Thank you for your sharing. I am worried that I lack creative ideas. It is your article that makes me full of hope. Thank you. But, I have a question, can you help me? https://accounts.binance.com/es-MX/register?ref=JHQQKNKN
Way cool! Some very valid points! I appreciate you writing this article plus the rest of the site is very good.
I really love your site.. Pleasant colors & theme. Did you develop this web site yourself? Please reply back as I’m trying to create my own blog and would love to find out where you got this from or just what the theme is called. Appreciate it.
En sevdiğim slot oyunu Thunderstruck II, her zaman büyük kazançlar sağlıyorum!
After I originally commented I seem to have clicked the -Notify me when new comments are added- checkbox and now each time a comment is added I receive four emails with the exact same comment. Perhaps there is a way you can remove me from that service? Many thanks.
An impressive share! I have just forwarded this onto a coworker who was conducting a little research on this. And he actually bought me dinner because I found it for him… lol. So let me reword this…. Thanks for the meal!! But yeah, thanx for spending time to talk about this matter here on your blog.
Excellent site you have here.. It’s hard to find high-quality writing like yours these days. I seriously appreciate people like you! Take care!!
Having read this I believed it was really informative. I appreciate you taking the time and effort to put this article together. I once again find myself personally spending way too much time both reading and leaving comments. But so what, it was still worth it.
Hi there, I do believe your site could possibly be having web browser compatibility issues. Whenever I look at your web site in Safari, it looks fine however, if opening in Internet Explorer, it has some overlapping issues. I simply wanted to give you a quick heads up! Apart from that, excellent site.
Oh my goodness! Impressive article dude! Thanks, However I am experiencing troubles with your RSS. I don’t know why I am unable to subscribe to it. Is there anyone else having the same RSS problems? Anyone that knows the answer will you kindly respond? Thanks.
Very good post! We will be linking to this particularly great post on our website. Keep up the good writing.
https://www.tellern.com Telegram应用是开源的,Telegram下载的程序支持可重现的构建。Telegram同时适用于以下环境:Android安卓端,iPhone 和 iPad及MacOS的Apple端,Windows/Mac/Linux桌面版
Oh my goodness! Impressive article dude! Thanks, However I am going through problems with your RSS. I don’t know the reason why I cannot subscribe to it. Is there anybody getting the same RSS problems? Anyone who knows the answer can you kindly respond? Thanx!
This is a topic that’s close to my heart… Thank you! Where are your contact details though?
Nice post. I learn something totally new and challenging on sites I stumbleupon on a daily basis. It’s always helpful to read through content from other writers and use a little something from their web sites.
I could not resist commenting. Very well written.
Having read this I thought it was really enlightening. I appreciate you finding the time and energy to put this content together. I once again find myself personally spending way too much time both reading and posting comments. But so what, it was still worthwhile.
I seriously love your blog.. Great colors & theme. Did you create this website yourself? Please reply back as I’m hoping to create my own personal site and would love to find out where you got this from or just what the theme is named. Thanks.
Hello there! This post couldn’t be written any better! Reading through this post reminds me of my previous roommate! He continually kept preaching about this. I most certainly will send this article to him. Fairly certain he will have a very good read. I appreciate you for sharing!
I was very happy to find this great site. I wanted to thank you for your time for this wonderful read!! I definitely loved every part of it and I have you saved as a favorite to look at new things on your site.
This is a very good tip especially to those fresh to the blogosphere. Short but very accurate info… Many thanks for sharing this one. A must read post!
Incorporating Sugar Defender right into my everyday program
overall wellness. As a person that focuses on healthy and balanced consuming,
I appreciate the added security this supplement provides.
Because beginning to take it, I’ve discovered a significant improvement in my power levels and a considerable reduction in my
wish for unhealthy snacks such a such an extensive impact
on my daily life.
A fascinating discussion is worth comment. I do believe that you need to publish more on this subject matter, it might not be a taboo matter but usually people don’t discuss such topics. To the next! Kind regards!
Deneme bonusu ile crash oyunlarına, özellikle Aviator’a sıfır yatırımla başlamak harika bir fırsat!
Greetings! Very useful advice in this particular post! It’s the little changes that produce the most significant changes. Thanks for sharing!
Slot oyunları çeşitliliği çok fazla, her oyuncuya hitap eden seçenekler var.
Your style is very unique compared to other people I have read stuff from. I appreciate you for posting when you’ve got the opportunity, Guess I will just bookmark this page.
Futbol bahislerinde strateji yaparak her maçta ekstra kazanç elde ediyorum!
Hi! Do you know if they make any plugins to help with SEO?
I’m trying to get my blog to rank for some targeted keywords but I’m not seeing
very good gains. If you know of any please share. Appreciate it!
I saw similar blog here: Warm blankets
Right here is the right website for anybody who would like to understand this topic. You understand a whole lot its almost tough to argue with you (not that I really will need to…HaHa). You definitely put a fresh spin on a topic that has been written about for decades. Great stuff, just excellent.
This website truly has all the info I wanted about this subject and didn’t know who to ask.
Everything is very open with a really clear explanation of the issues. It was really informative. Your website is very helpful. Many thanks for sharing!
Bu casino’da bahis yaparken strateji yapmak her zaman kazançlı çıkmamı sağlıyor.
Greetings! Very helpful advice in this particular post! It’s the little changes that produce the largest changes. Many thanks for sharing!
Good blog post. I certainly appreciate this site. Thanks!
You ought to be a part of a contest for one of the greatest websites online. I will highly recommend this blog!
Casino’nun sunduğu bonuslar sayesinde slot oyunlarında daha fazla kazanma şansı buluyorum.
Deneme bonusu sayesinde hiç para yatırmadan kazanç sağlamaya başladım, çok memnunum!
Aviator gibi crash oyunlarını deneme bonusu ile oynamak, risksiz kazanç sağlamanın en kolay yolu!
Futbol bahislerinde aldığım bonuslarla daha fazla maç tahmini yapabiliyorum ve kazancımı artırıyorum!
Spot on with this write-up, I seriously feel this website needs a lot more attention. I’ll probably be back again to see more, thanks for the info!
Bu casino’da futbol bahislerinde doğru stratejiyle sürekli kazanç elde ediyorum!
Good web site you have got here.. It’s hard to find good quality writing like yours these days. I honestly appreciate people like you! Take care!!
An impressive share! I have just forwarded this onto a friend who was conducting a little research on this. And he actually ordered me lunch simply because I found it for him… lol. So allow me to reword this…. Thank YOU for the meal!! But yeah, thanx for spending time to discuss this issue here on your site.
I blog frequently and I really thank you for your content. The article has really peaked my interest. I will book mark your site and keep checking for new information about once per week. I opted in for your Feed as well.
Howdy! I could have sworn I’ve been to this site before but after looking at some of the articles I realized it’s new to me. Anyhow, I’m definitely delighted I found it and I’ll be bookmarking it and checking back often!
It’s difficult to find educated people for this topic, but you sound like you know what you’re talking about! Thanks
Hi, I do believe this is an excellent web site. I stumbledupon it 😉 I may revisit yet again since i have bookmarked it. Money and freedom is the best way to change, may you be rich and continue to guide other people.
This web site really has all of the information and facts I needed about this subject and didn’t know who to ask.
Deneme bonusu sayesinde hiç para kaybetmeden büyük kazançlar sağladım, çok memnunum!
Next time I read a blog, I hope that it won’t disappoint me just as much as this particular one. I mean, I know it was my choice to read, nonetheless I actually thought you’d have something helpful to talk about. All I hear is a bunch of moaning about something you could fix if you were not too busy seeking attention.
Greetings! Very useful advice in this particular post! It is the little changes that will make the largest changes. Thanks a lot for sharing!
Excellent site you have got here.. It’s hard to find high quality writing like yours nowadays. I truly appreciate individuals like you! Take care!!
Right here is the right webpage for anyone who wants to understand this topic. You understand a whole lot its almost tough to argue with you (not that I actually would want to…HaHa). You certainly put a fresh spin on a subject that’s been written about for ages. Wonderful stuff, just excellent.
Can I simply just say what a relief to find someone that truly knows what they’re discussing on the net. You actually understand how to bring an issue to light and make it important. More people need to look at this and understand this side of the story. I was surprised you’re not more popular because you surely possess the gift.
Howdy! I could have sworn I’ve been to your blog before but after browsing through some of the articles I realized it’s new to me. Nonetheless, I’m definitely pleased I came across it and I’ll be book-marking it and checking back often.
En sevdiğim takımın maçlarına bahis yaparken kazanç fırsatları hiç bitmiyor!
I want to to thank you for this excellent read!! I definitely loved every little bit of it. I have got you bookmarked to check out new things you post…
Bu sitenin sunduğu deneme bonusu ile bedava Aviator oynayarak büyük ödüller kazandım!
Hızlı ödeme seçenekleri sayesinde anında kazancımı çekebiliyorum.
Bahis siteleri deneme bonusu ile oyunlara riske girmeden başlayarak hemen kazanç sağladım!
It’s hard to come by well-informed people for this subject, however, you seem like you know what you’re talking about! Thanks
Düşük bahislerle uzun süre oynamak Razor Shark’ta kazançlarımı artırıyor.
Bu bahis sitesinin sunduğu deneme bonusu ile crash oyunlarında, özellikle Aviator’da, büyük kazançlar sağladım!
Bu casino’daki slot oyunları ile büyük jackpot kazançlarına ulaşmak çok heyecan verici!
Yüksek bahislerle futbol bahislerinde devasa ödüller kazanmak çok kolay!
Crash oyunları, özellikle Aviator, deneme bonusu ile hiç yatırım yapmadan oynanabilecek en heyecan verici oyunlar!
Deneme bonusu ile ilk bahislerimde hiç para kaybetmeden kazandım, çok heyecanlı!
Deneme bonusu ile bahis yaparken hiç risk almadan kazanmaya başladım!
Casino’da slot oynarken kazandığım büyük ödüller sayesinde mutluluğum tarif edilemez!
Bu sitenin sunduğu deneme bonusu ile hemen kazanmaya başlamak gerçekten çok kolay!
Casino’nun sunduğu promosyonlar futbol bahislerinde her zaman ekstra avantaj sağlıyor!
En sevdiğim slot oyunu Wild West Gold, her zaman büyük kazançlar elde ediyorum!
Yüksek bahislerle futbol bahislerinde devasa ödüller kazanmak çok heyecan verici!
Casino’nun sunduğu promosyonlar futbol bahislerinde ekstra avantajlar sağlıyor, her maç daha kazançlı!
Bu casino’daki slot oyunlarında kazanmak gerçekten çok kolay ve eğlenceli.
Futbol bahislerimde aldığım bonuslar ile daha fazla bahis yapabiliyorum ve kazancımı artırıyorum!
Casino’nun sunduğu promosyonlar futbol bahislerinde her zaman ekstra kazanç sağlıyor!
Bu casino’da oynadığım slot oyunları sayesinde büyük kazançlar elde ediyorum.
Casino’nun sunduğu geniş bahis seçenekleri ile her futbol maçında kazanç fırsatları buluyorum.
WPS官网下载WPS Office: 一站式办公服务平台: 新升级,无广告,AI办公更高效. 立即下载. 登录使用. WPS 365: 面向组织和企业的WPS 365: 一站式AI办公,生产力即刻起飞. 了解更多. 咨询,记忆体占用低,体积轻运行快. 将文字、表格、演示、PDF等融合为一个组件。
HelloWord翻译Hello World聊天翻译助手专注于为出海企业提供高质量的即时聊天翻译服务,专业聊天翻译技术,极速稳定收发,全球畅游,使用邮箱免费注册登录体验,专业翻译技术团队开发,超数百家企业信赖,支持whatsapp Line Tinder Twitter Instagram Telegram Zalo Facebook Badoo Bumble Quora Linkedin googleVoice Crisp Hangouts TextNow VK等软件的实时聊天翻译,无限网页多开。支持facebook群发,whastsapp群发,googleVoice群发
Casino’nun sunduğu slot oyunları her zaman heyecan dolu ve kazanç fırsatları sunuyor.
Deneme bonusu ile bahis yaparken hiçbir risk almadan kazanç fırsatlarını değerlendirdim.
HelloWord翻译Hello World聊天翻译助手专注于为出海企业提供高质量的即时聊天翻译服务,专业聊天翻译技术,极速稳定收发,全球畅游,使用邮箱免费注册登录体验,专业翻译技术团队开发,超数百家企业信赖,支持whatsapp Line Tinder Twitter Instagram Telegram Zalo Facebook Badoo Bumble Quora Linkedin googleVoice Crisp Hangouts TextNow VK等软件的实时聊天翻译,无限网页多开。支持facebook群发,whastsapp群发,googleVoice群发
Hi, I do think this is a great blog. I stumbledupon it 😉 I’m going to revisit once again since i have book-marked it. Money and freedom is the greatest way to change, may you be rich and continue to guide others.
Bu casino’nun sunduğu slot oyunları gerçekten büyük kazanç fırsatları sunuyor.
VIP programı çok avantajlı, sadık oyunculara özel bonuslar sunuyorlar.
Bu casino’daki geniş bahis seçenekleri sayesinde her seviyede oyuncu kazanma şansı yakalıyor.
Casino’nun sunduğu turnuvalar sayesinde slot oyunlarında ekstra ödüller kazanmak çok kolay.
En sevdiğim slot oyunu Book of Ra, grafikler harika ve kazançlar büyük!
gluco6 reviews : https://gluco6reviews.usaloves.com/
I used to be able to find good info from your blog articles.
gluco6 reviews : https://gluco6reviews.usaloves.com/
Greetings! Very useful advice in this particular article! It’s the little changes that make the most significant changes. Thanks a lot for sharing!
Spot on with this write-up, I truly believe this amazing site needs much more attention. I’ll probably be returning to read more, thanks for the information!
I used to be able to find good information from your content.
有道词典是由网易有道出品的全球首款基于搜索引擎技术的全能免费语言翻译软件。简介. 支持中文、英语、日语、韩语、法语、德语、俄语、西班牙语、葡萄牙语、藏语、西语等109种语言翻译。拍照翻译、语音翻译、对话翻译、在线翻译、离线翻译更顺畅。更多的翻译 https://www.youdaoo.com
HelloWord翻译Hello World聊天翻译助手专注于为出海企业提供高质量的即时聊天翻译服务,专业聊天翻译技术,极速稳定收发,全球畅游,使用邮箱免费注册登录体验,专业翻译技术团队开发,超数百家企业信赖,支持whatsapp Line Tinder Twitter Instagram Telegram Zalo Facebook Badoo Bumble Quora Linkedin googleVoice Crisp Hangouts TextNow VK等软件的实时聊天翻译,无限网页多开。支持facebook群发,whastsapp群发,googleVoice群发
Bu casino’daki futbol bahis seçenekleri, her maçta kazanma şansımı artırıyor!
Way cool! Some extremely valid points! I appreciate you penning this post and also the rest of the website is also really good.
Canlı casino oyunları ve slotların birleşimi, harika bir oyun deneyimi sunuyor.
I wanted to thank you for this good read!! I absolutely loved every bit of it. I have you book marked to look at new stuff you post…
WPS官网下载WPS Office: 一站式办公服务平台: 新升级,无广告,AI办公更高效. 立即下载. 登录使用. WPS 365: 面向组织和企业的WPS 365: 一站式AI办公,生产力即刻起飞. 了解更多. 咨询,记忆体占用低,体积轻运行快. 将文字、表格、演示、PDF等融合为一个组件。
Deneme bonusu ile bahis yaparken riske girmeden kazanç sağladım!
Oh my goodness! Awesome article dude! Thanks, However I am encountering difficulties with your RSS. I don’t understand why I am unable to join it. Is there anyone else having identical RSS problems? Anyone that knows the answer can you kindly respond? Thanks!!
Casino’nun sunduğu jackpot slot oyunları ile büyük ödüller kazanmak mümkün.
En sevdiğim slot oyunu Vikings Go Berzerk, büyük kazançlar elde ediyorum!
Bahis sitelerinin sunduğu deneme bonusları ile bedava kazanç elde etmek çok kolay!
En sevdiğim slot oyunu Sweet Bonanza, her spin büyük bir kazanç fırsatı sunuyor.
HelloWord翻译Hello World聊天翻译助手专注于为出海企业提供高质量的即时聊天翻译服务,专业聊天翻译技术,极速稳定收发,全球畅游,使用邮箱免费注册登录体验,专业翻译技术团队开发,超数百家企业信赖,支持whatsapp Line Tinder Twitter Instagram Telegram Zalo Facebook Badoo Bumble Quora Linkedin googleVoice Crisp Hangouts TextNow VK等软件的实时聊天翻译,无限网页多开。支持facebook群发,whastsapp群发,googleVoice群发
I really love your site.. Very nice colors & theme. Did you develop this amazing site yourself? Please reply back as I’m planning to create my own personal website and want to find out where you got this from or exactly what the theme is named. Kudos.
Bu casino’nun sunduğu geniş bahis seçenekleri futbol maçlarına ekstra heyecan katıyor!
Greetings! Very useful advice in this particular post! It is the little changes that will make the biggest changes. Thanks a lot for sharing!
En sevdiğim slot oyunu Great Rhino Megaways, her spin büyük bir kazanç fırsatı sunuyor.
Good post. I learn something new and challenging on websites I stumbleupon on a daily basis. It will always be interesting to read content from other authors and use something from other web sites.
Casino’nun sunduğu promosyonlar ve bonuslar ile her gün slot oyunlarında kazanıyorum.
Casino bonusları ile slot oyunlarında daha fazla kazanıyorum, fırsatlar harika!
Right here is the right website for anyone who would like to understand this topic. You realize a whole lot its almost hard to argue with you (not that I actually will need to…HaHa). You definitely put a brand new spin on a topic which has been written about for ages. Wonderful stuff, just great.
Aw, this was an incredibly good post. Taking a few minutes and actual effort to create a really good article… but what can I say… I hesitate a whole lot and never manage to get anything done.
HelloWord翻译Hello World聊天翻译助手专注于为出海企业提供高质量的即时聊天翻译服务,专业聊天翻译技术,极速稳定收发,全球畅游,使用邮箱免费注册登录体验,专业翻译技术团队开发,超数百家企业信赖,支持whatsapp Line Tinder Twitter Instagram Telegram Zalo Facebook Badoo Bumble Quora Linkedin googleVoice Crisp Hangouts TextNow VK等软件的实时聊天翻译,无限网页多开。支持facebook群发,whastsapp群发,googleVoice群发
This is a really good tip especially to those new to the blogosphere. Short but very precise information… Thanks for sharing this one. A must read post!
Hi, I do think your website may be having browser compatibility problems. Whenever I look at your website in Safari, it looks fine but when opening in IE, it has some overlapping issues. I merely wanted to give you a quick heads up! Apart from that, excellent blog!
Casino’nun sunduğu promosyonlar ve bonuslar ile her gün slot oyunlarında kazanıyorum.
I could not resist commenting. Perfectly written.
HelloWord翻译Hello World聊天翻译助手专注于为出海企业提供高质量的即时聊天翻译服务,专业聊天翻译技术,极速稳定收发,全球畅游,使用邮箱免费注册登录体验,专业翻译技术团队开发,超数百家企业信赖,支持whatsapp Line Tinder Twitter Instagram Telegram Zalo Facebook Badoo Bumble Quora Linkedin googleVoice Crisp Hangouts TextNow VK等软件的实时聊天翻译,无限网页多开。支持facebook群发,whastsapp群发,googleVoice群发
Everything is very open with a very clear description of the issues. It was truly informative. Your site is extremely helpful. Thank you for sharing!
It’s hard to find experienced people for this topic, but you sound like you know what you’re talking about! Thanks
I was able to find good advice from your blog posts.
En sevdiğim slot oyunu Book of Dead, her spin büyük bir heyecan ve kazanç sağlıyor.
HelloWord翻译Hello World聊天翻译助手专注于为出海企业提供高质量的即时聊天翻译服务,专业聊天翻译技术,极速稳定收发,全球畅游,使用邮箱免费注册登录体验,专业翻译技术团队开发,超数百家企业信赖,支持whatsapp Line Tinder Twitter Instagram Telegram Zalo Facebook Badoo Bumble Quora Linkedin googleVoice Crisp Hangouts TextNow VK等软件的实时聊天翻译,无限网页多开。支持facebook群发,whastsapp群发,googleVoice群发
Hi! I could have sworn I’ve been to this web site before but after going through some of the articles I realized it’s new to me. Regardless, I’m certainly delighted I came across it and I’ll be book-marking it and checking back frequently!
Bahis sitelerinde deneme bonusu ile oyunlara bedava başlayarak kazanabilirsiniz!
在这里下载Telegram官网最新版 ,适用于所有主流操作系统。本站为你提供详细的纸飞机使用指南,包括如何下载、安装以及设置中文界面,帮助你轻松使用这一全球领先的通讯 https://www.tellern.com
有道词典是由网易有道出品的全球首款基于搜索引擎技术的全能免费语言翻译软件。简介. 支持中文、英语、日语、韩语、法语、德语、俄语、西班牙语、葡萄牙语、藏语、西语等109种语言翻译。拍照翻译、语音翻译、对话翻译、在线翻译、离线翻译更顺畅。更多的翻译 https://www.youdaoo.com
WPS Office: 一站式办公服务平台: 新升级,无广告,AI办公更高效. 立即下载. 登录使用. WPS 365: 面向组织和企业的WPS 365: 一站式AI办公,生产力即刻起飞. 了解更多. 咨询,记忆体占用低,体积轻运行快. 将文字、表格、演示、PDF等融合为一个组件。WPS下载 https://www.wpsue.com
Hello World聊天翻译助手专注于为出海企业提供高质量的即时聊天翻译服务,专业聊天翻译技术,极速稳定收发,全球畅游,使用邮箱免费注册登录体验,专业翻译技术团队开发,超数百家企业信赖,支持whatsapp Line Tinder Twitter Instagram Telegram Zalo Facebook Badoo Bumble Quora Linkedin googleVoice Crisp Hangouts TextNow VK等软件的实时聊天翻译,无限网页多开。支持facebook群发,whastsapp群发,googleVoice群发 HelloWord翻译 https://www.hiword.cc
Telegram应用是开源的,Telegram官网下载 https://www.telegramv.net 的程序支持可重现的构建。Telegram同时适用于以下环境:Android安卓端,iPhone 和 iPad及MacOS的Apple端,Windows/Mac/Linux桌面版
Oh my goodness! Impressive article dude! Thank you so much, However I am having troubles with your RSS. I don’t know why I am unable to join it. Is there anybody having identical RSS issues? Anyone that knows the solution can you kindly respond? Thanx!!
https://www.telqq.com Telegram群组,Telegram群组导航。收录Telegram上的优质频道和群组,打造一个高质量Telegram导航。TGNAV收录整理了Telegram上的许多优质频道、群组、机器人,帮助用户发现更多优质的群组。
https://www.tellern.com Telegram应用是开源的,Telegram下载的程序支持可重现的构建。Telegram同时适用于以下环境:Android安卓端,iPhone 和 iPad及MacOS的Apple端,Windows/Mac/Linux桌面版
Your point of view caught my eye and was very interesting. Thanks. I have a question for you.
Casino’da slot oyunlarının çeşitliliği inanılmaz, her zevke uygun bir oyun var!
Yüksek bahislerle oynadığım futbol maçlarında devasa ödüller kazanıyorum!
Casino’nun sunduğu promosyonlar ile slot oyunlarında daha fazla kazanç elde ediyorum.
En sevdiğim slot oyunu Fire in the Hole, her spin heyecan verici ve kazanç dolu.
En sevdiğim slot oyunu Great Rhino, her spin büyük bir heyecan ve kazanç sağlıyor.
Bahis siteleri deneme bonusu ile crash oyunlarını oynamak, özellikle Aviator’da, büyük kazanç fırsatları sunuyor!
Bu casino’da oynadığım slot oyunları ile kazançlarım her geçen gün artıyor.
En sevdiğim slot oyunu Bonanza, her spin büyük bir heyecan sunuyor.
Bu casino’nun futbol bahis seçenekleri çok geniş, her maç için farklı bahis stratejileri deniyorum!